.png)
Now we can predict the shape of the molecule by first finding the total electron pairs around the central atom using the formula, Lone Pair- Lone Pair (LP – LP) > Lone Pair- Bond Pair (LP – BP) > Bond Pair- Bond Pair (BP – BP) The force of repulsion between Lone Pair- Lone Pair, Lone Pair- Bond Pair, Bond Pair- Bond Pair (BP – BP) is in the order mentioned below: The molecule in a linear shape is arranged in such a way that it has two valance shells and the bond pair in this arrangement are arranged in such a manner that their repulsion is minimum, this is achieved by taking the bond pair in opposite directions.īeF 2 is an example of a Linear Shape Molecule. The various shape of the molecules are discussed in the article below, Linear Shape of Molecule Various Shapes of molecules according to the VSEP number is tabulated below: We easily find the VESP number by taking the sum of the Lone Pairs and Bond Pairs of the molecule. The VSEP number of the molecule is the number that describes the shape of the molecule. The lone pair and the pair are sums to find the VESP number of the molecule which is used to explain the shape of the molecule.The electrons shared by other atoms with the central atom are accounted as the bond pair of electrons.The lone pairs of the central atom are counted by taking the central atom’s outermost shell into account.This atom can easily share its electron with other atoms of the molecule. The least electronegative element of the molecule must be chosen as the central element.We can easily predict the shape of the molecules using the VESPR theory and the rules we must follow to predict the shape of the molecule are, 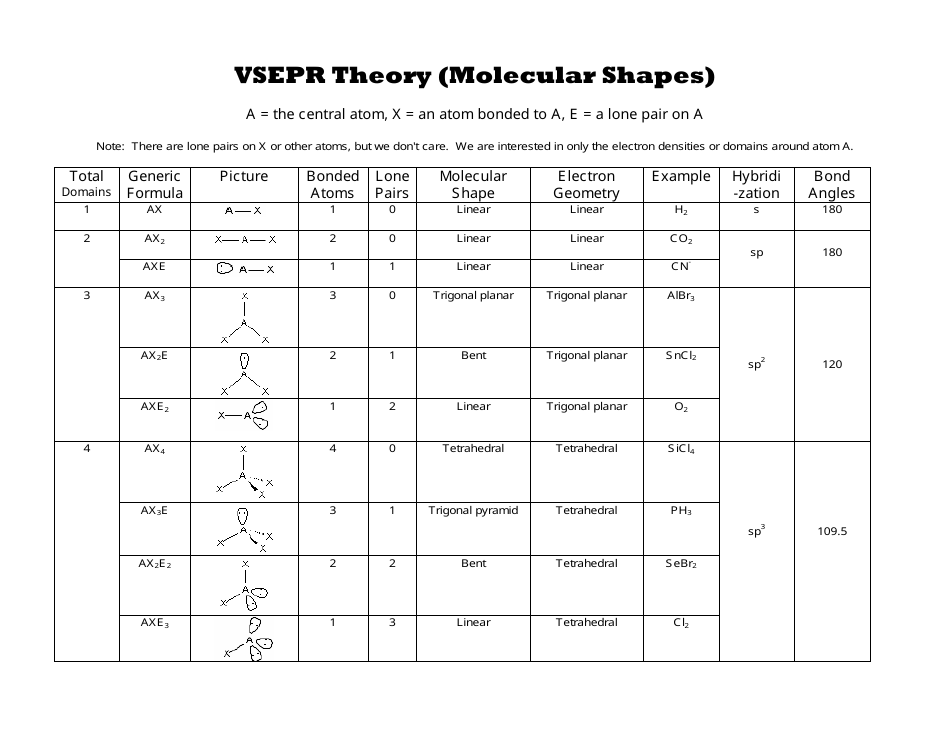
Lone Pair – Lone Pair > Lone Pair – Bond Pair > Bond Pair – Bond Pair Predicting Shapes of Molecules Using VSEPR Theory The force of repulsion between the lone pairs, lone pair, and bond pair, and bond pairs follows the order.In each resonance state of the molecule, their structure is explained using the VSEPR theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
